Chlorophyll Fluorescence Measuring Methods OJIP - part 2
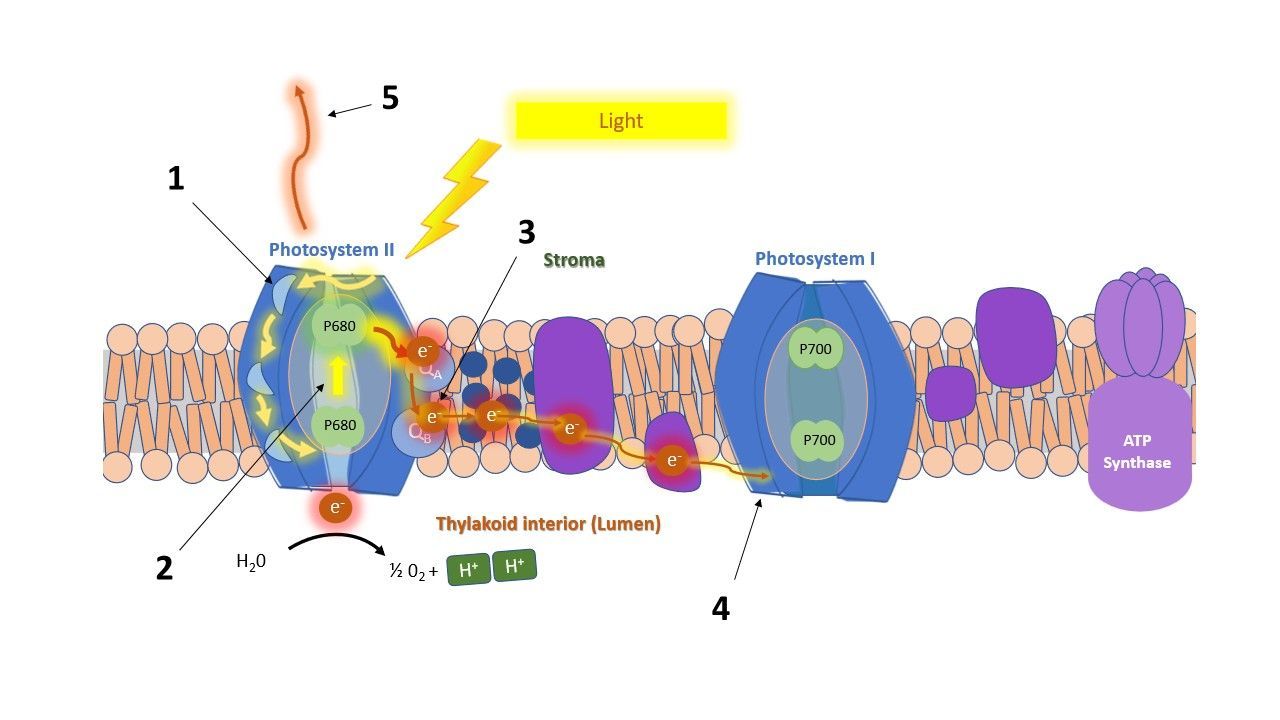
OJIP is a measurement method used to study chlorophyll fluorescence kinetics in detail, specifically the rapid initial rise in fluorescence during a saturating light pulse. This rise reveals information about energy fluxes and electron transport within the photosynthetic apparatus.
In Part 1 of our OJIP blog series, I explained which physiological processes cause this characteristic fluorescence increase. In Part 2, I will focus on the parameters that can be derived from the OJIP transient (JIP test). These parameters allow us to quantify the processes discussed earlier, turning the curve shape into measurable physiological indicators.
- Schematic overview
JIP parameters focus on the pathway of energy through the photosynthetic system: they track the fate of light energy from the moment it is absorbed, showing whether it is used to drive photosynthesis or dissipated as heat or fluorescence. The parameters can be grouped into three categories: energy fluxes, quantum yields and efficiencies.
1.1. Energy fluxes
Flux parameters describe the amount of energy flow through the different steps of the electron transport chain:
(1) Absorption flux: Photons absorbed by the antenna pigments and creating excited chlorophyll.
(2) Trapping flux: Channeled energy from excited chlorophyll to the reaction center to be converted into the electron transport chain (QA reduction).
(3) Electron flux: Electron transport further than QA
(4) Reduction flux: Reduction of end electron acceptors at the PSI side of the electron transport chain.
(5) Dissipation flux: Energy that is dissipated as heat or fluorescence.
1.2. Quantum yields + efficiencies
Next to the energy fluxes, we can calculate the fraction of absorbed energy by PSII that is used for a specific photochemical event. These fractions are also known as the quantum yield. Furthermore, the efficiency of the the event (with trapped energy) can also be calculated:
- Quantum yield of primary photochemistry, reducing QA (φP₀, often expressed as Fv/Fm)
- Quantum yield of electron transport from QA → QB → PQ pool (φE₀)
- Quantum yield of electron transport to the final PSI acceptors (φR₀)
- Efficiency with which a trapped excitation moves an electron further than QA into the electron transport chain (ΨE₀)
- Efficiency with which electrons arriving at PSI reduce the end acceptors FA/FB, ferredoxin, or NADP⁺ (δR₀)
These parameters provide insight into the functioning of the photosynthetic apparatus and are valuable for studying, for example, the mode of action of various compounds or the effects of environmental stresses.
A parameter that combines multiple of these processes, and that is highly sensitive to stress is the Performance Index (PI).
1.3. Performance index
The Performance Index (PI) was introduced because the commonly used parameter for photosynthetic efficiency, Fv/Fm, is not always sensitive enough to detect early or subtle stress responses. PI integrates multiple components of the photosynthetic process into a single value (energy absorption, trapping and electron transport). It is widely used by plant physiologists to evaluate the effects of biotic and abiotic stresses, and it is also a powerful parameter for breeding programs to rapidly screen genotypes under conditions such as drought or salinity stress. PI is considered one of the most sensitive OJIP-derived parameters.
There are two Performance Index variants: PI_ABS and PI_TOTAL.
PI_ABS combines three components into a single value:
- The density of active reaction centres
- The efficiency of trapping (the probability that absorbed light energy closes QA)
- The efficiency of electron transport beyond QA
PI_TOTAL
includes the three components of PI_ABS and adds a fourth:
4. The efficiency of electron transport to the final PSI acceptors
Because PI incorporates multiple steps in the photosynthetic electron transport chain, any disturbance affecting one of the components will be reflected in the PI value, making it more sensitive than individual parameters alone.
2. Literature review
To provide an overview of the JIP parameters and how they can be used, we performed a literature survey to assess how different types of stresses affect JIP parameters. The following stresses were included: salt stress, drought stress, heat stress, cold stress, and high-light stress.
In general, several JIP parameters consistently respond to stress across studies, but the direction of the response can vary: some parameters increase under stress in a certain species or conditions, while others decrease. The exact pattern depends on the plant species, or even cultivar, and the severity of the stress.
A table overview of the results is summarized below (click the link).
3. How parameters from OJIP are often visualized and interpreted
The large number of available OJIP parameters can make interpretation challenging. However, it is valuable for researchers to view the full set of parameters, as each one reflects a different component of the photosynthetic apparatus. Effects in one part of the electron transport chain may not appear in another, so examining multiple parameters can reveal where specific changes or stress responses occur.
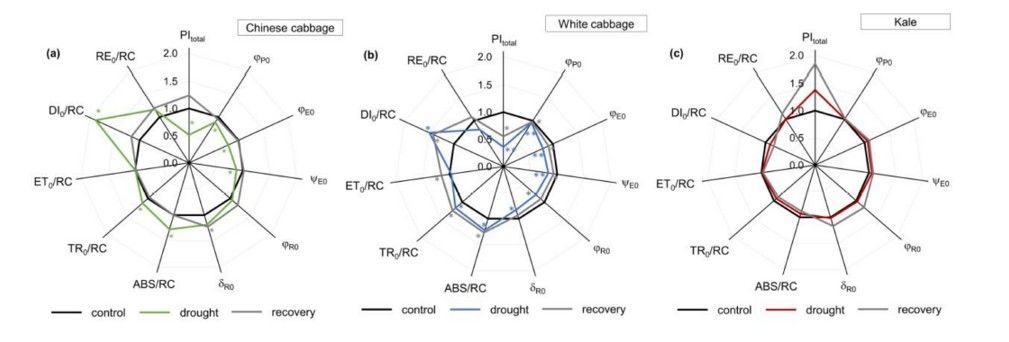
A useful way to visualize the complete dataset is through spider plots (Figure 1). These plots allow many parameters to be displayed simultaneously, making it easier to identify patterns, compare treatments, or detect stress-induced deviations.
In some studies, authors visualize the different energy fluxes (per cross‑section) using a leaf model, such as the one shown below (Figure 2). In this type of schematic, the width of the arrows represents the magnitude of each energy flux, with yellow indicating absorption flux, dark blue representing electron transport flux, light blue showing trapping flux, and red depicting dissipation flux. The black dots inside the central square illustrate the number of inactive or silent reaction centres.
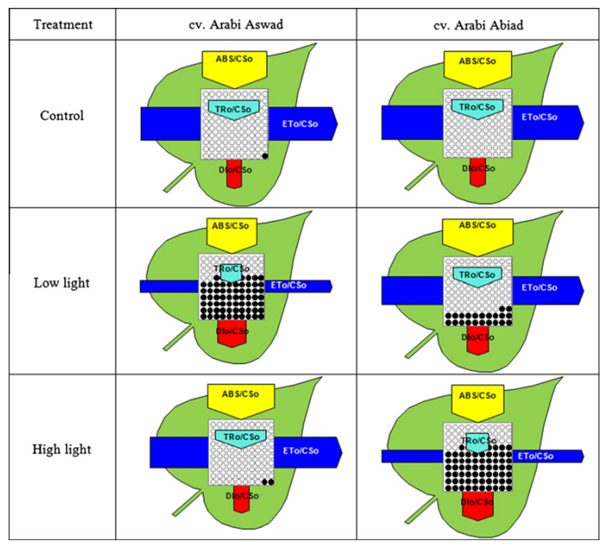
This type of model is highly informative, as it summarizes the state of the photosynthetic apparatus in a single image. In the example shown above, two barley cultivars were compared under high‑light and low light conditions. From the flux pattern, it becomes clear that the cultivar ‘Arabi Aswad’ (left) is more capable of handling high light, having a larger electron transport flux, lower dissipation flux, and more active reaction centers. The cultivar ‘Arabi Abiad’ (right) is better adapted to low light.
The model can also be expressed as a membrane model, showing the fluxes per reaction center (RC). An example is given below (Figure 3).
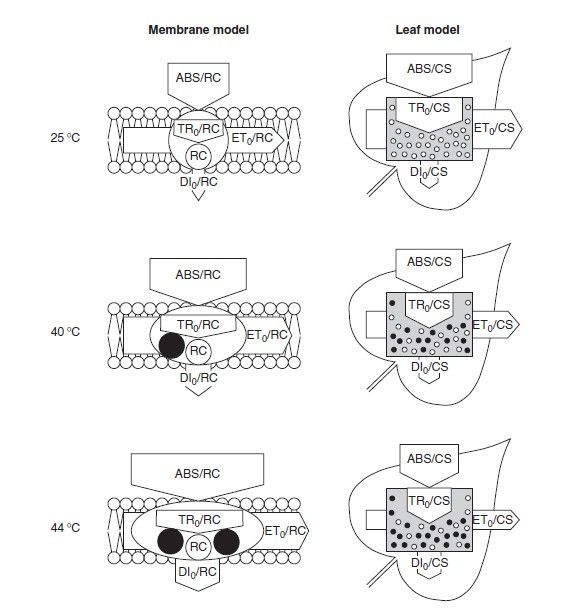
Here, the effect of heating on the membrane model (per reaction center) is described by Strasser as:
• "Absorption per active reaction center increases due to the inactivation of some RCs"
• "The ratio of total dissipation to the amount of active RCs increases due to the high dissipation of the inactive RCs"
• "Electron transport per active reaction center increases due to a thermal activation of the dark reactions"
And on the leaf model (per cross section) as:
• "Decrease of electron transport per excited cross section due to the inactivation of reaction center complexes"
• "Decrease of the density of active reaction centers RC/CS (indicated as open circles)
• "Increase of the energy dissipation per excited cross section."
• "Decrease of the energy absorbed per excited cross section ABS/CS."
4. References
1. Strasser, R.J., Srivastava, A. And Govindjee (1995). Polyphasic chlorophyll a fluorescence transient in plants and cyanobacteria. Photochemistry and Photobiology 61: 32-42.
2. Strasser, R.J., Srivastava, A. And Tsimilli-Michael, M. (2000). The fluorescence transient as a tool to characterize and screen photosynthetic samples. In: Ynus, M. (Ed.) Probing Photosynthesis: Mechanisms, Regulation and Adaptation. Taylor and Francis, London: 445-483.
3. Strasser, R.J., Tsimilli-Micael, M. and Srivastava, A. (2004). Analysis of the Chlorophyll a fluorescence transient. In: Papageorgiou, G.C. and Govindjee (Eds.) Chlorophyll a fluorescence: a signature of photosynthesis. Springer, Dordrecht: 321-362.
4. Lotfi, R., H. M. Kalaji, G. R. Valizadeh, E. Khalilvand Behrozyar, A. Hemati, P. Gharavi‑Kochebagh, and A. Ghassemi. 2018. Effects of humic acid on photosynthetic efficiency of rapeseed plants growing under different watering conditions. Photosynthetica 56(3): 962–970. https://doi.org/10.1007/s11099-017-0745-9.
5. Zushi, K., S. Kajiwara, and N. Matsuzoe. 2012. Chlorophyll a fluorescence OJIP transient as a tool to characterize and evaluate response to heat and chilling stress in tomato leaf and fruit. Scientia Horticulturae 148: 39–46. https://doi.org/10.1016/j.scienta.2012.09.022.
6. Kalaji, H. M., A. Rastogi, M. Živčák, M. Brestič, A. Daszkowska‑Golec, K. Sitko, K. Y. Alsharafa, R. Lotfi, P. Stypiński, I. A. Samborska, and M. D. Cetner. 2018. Prompt chlorophyll fluorescence as a tool for crop phenotyping: an example of barley landraces exposed to various abiotic stress factors. Photosynthetica 56: 953–961. https://doi.org/10.1007/s11099-018-0766-z.
7. Chen, X., Y. Zhou, Y. Cong, P. Zhu, J. Xing, J. Cui, W. Xu, Q. Shi, M. Diao, and H.‑Y. Liu. 2021. Ascorbic acid‑induced photosynthetic adaptability of processing tomatoes to salt stress probed by fast OJIP fluorescence rise. Frontiers in Plant Science 12: 594400. https://doi.org/10.3389/fpls.2021.594400.
9. Stefanov, D., V. Petkova, and I. D. Denev. 2011. Screening for heat tolerance in common bean (Phaseolus vulgaris L.) lines and cultivars using JIP‑test. Scientia Horticulturae 128(1): 1–6. https://doi.org/10.1016/j.scienta.2010.12.003.
11. Jedmowski, C., and W. Brüggemann. 2015. Imaging of fast chlorophyll fluorescence induction curve (OJIP) parameters, applied in a screening study with wild barley (Hordeum spontaneum) genotypes under heat stress. Journal of Photochemistry and Photobiology B: Biology 151: 153–160. https://doi.org/10.1016/j.jphotobiol.2015.07.020.
13. Kalaji, H. M., A. Oukarroum, V. Alexandrov, M. Kouzmanova, M. Brestic, M. Zivcak, I. A. Samborska, M. D. Cetner, S. I. Allakhverdiev, and V. Goltsev. 2014. Identification of nutrient deficiency in maize and tomato plants by in vivo chlorophyll a fluorescence measurements. Plant Physiology and Biochemistry 81: 16–25. https://doi.org/10.1016/j.plaphy.2014.03.029.
14. Mehta, P., S. I. Allakhverdiev, and A. Jajoo. 2010. Characterization of photosystem II heterogeneity in response to high salt stress in wheat leaves (Triticum aestivum). Photosynthesis Research 105(3): 249–255. https://doi.org/10.1007/s11120-010-9588-y.
15. Jedmowski, C., A. Ashoub, and W. Brüggemann. 2013. Reactions of Egyptian landraces of Hordeum vulgare and Sorghum bicolor to drought stress, evaluated by the OJIP fluorescence transient analysis. Acta Physiologiae Plantarum 35: 345–354. https://doi.org/10.1007/s11738-012-1077-9.
16. Ren, J., P. Guo, X. Zhao, X. Ma, X. Ai, J. Wang, H. Zou, and H. Yu. 2025. Differential photosynthetic responses to drought stress in peanut varieties: insights from transcriptome profiling and JIP‑Test analysis. BMC Plant Biology 25: 957. https://doi.org/10.1186/s12870-025-06984-y.
17. Mihaljević, I., M. Viljevac Vuletić, V. Tomaš, Z. Zdunić, D. Vuković, and K. Dugalić. 2024. Assessment of photosynthetic capacity of two blackberry cultivars subjected to salt stress by the JIP fluorescence test. Journal of Berry Research 14(1): 1–13. https://doi.org/10.3233/JBR-230026.
18. Rastogi, A., M. Kovář, X. He, M. Živčák, S. Kataria, H. M. Kalaji, M. Skalicky, U. F. Ibrahimova, S. Hussain, S. Mbarki, and M. Brestic. 2020. JIP‑test as a tool to identify salinity tolerance in sweet sorghum genotypes. Photosynthetica 58(Special Issue): 518–528. https://doi.org/10.32615/ps.2019.169.
19. Ranjbarfordoei, A., R. Samson, and P. Van Damme. 2006. Chlorophyll fluorescence performance of sweet almond (Prunus dulcis (Miller) D. Webb) in response to salinity stress induced by NaCl. Photosynthetica 44(4): 513–522. https://doi.org/10.1007/s11099-006-0064-z.
20. Jafarinia, M., and M. Shariati. 2012. Effects of salt stress on photosystem II of canola plant (Brassica napus L.) probed by chlorophyll a fluorescence measurements. Iranian Journal of Science and Technology (Sciences) 36(1): 71–76. https://doi.org/10.22099/ijsts.2012.2058.
21. Zushi, K., and N. Matsuzoe. 2017. Using of chlorophyll a fluorescence OJIP transients for sensing salt stress in the leaves and fruits of tomato. Scientia Horticulturae 219: 216–221. https://doi.org/10.1016/j.scienta.2017.03.016.
22. Giorio, P., and M. H. Sellami. 2021. Polyphasic OKJIP chlorophyll a fluorescence transient in a landrace and a commercial cultivar of sweet pepper (Capsicum annuum L.) under long‑term salt stress. Plants 10(5): 887. https://doi.org/10.3390/plants10050887.
23. Akhter, M. S., S. Noreen, S. Mahmood, H.‑u‑R. Athar, M. Ashraf, A. A. Alsahli, and P. Ahmad. 2021. Influence of salinity stress on PSII in barley (Hordeum vulgare L.) genotypes, probed by chlorophyll‑a fluorescence. Journal of King Saud University – Science 33(1): 101239. https://doi.org/10.1016/j.jksus.2020.101239.
24. Zhang, T., H. Gong, X. Wen, and C. Lu. 2010. Salt stress induces a decrease in excitation energy transfer from phycobilisomes to photosystem II but an increase to photosystem I in the cyanobacterium Spirulina platensis. Journal of Plant Physiology 167(12): 951–958. https://doi.org/10.1016/j.jplph.2009.12.020.
25. Weng, H., M. Wu, X. Li, L. Wu, J. Li, T. O. Atoba, J. Zhao, R. Y. Wu, and D. Ye. 2023. High‑throughput phenotyping salt tolerance in JUNCAOs by combining prompt chlorophyll a fluorescence with hyperspectral spectroscopy. Plant Science 330: 111660. https://doi.org/10.1016/j.plantsci.2023.111660.
26. Chen, X., X. Liu, Y. Cong, Y. Jiang, J. Zhang, Q. Yang, and H. Liu. 2025. Melatonin alleviates photosynthetic injury in tomato seedlings subjected to salt stress via OJIP chlorophyll fluorescence kinetics. Plants 14(5): 824. https://doi.org/10.3390/plants14050824.
27. Antunović Dunić, J., Mlinarić, S., Pavlović, I., Lepeduš, H., and Salopek‑Sondi, B. 2023. Comparative analysis of primary photosynthetic reactions assessed by OJIP kinetics in three Brassica crops after drought and recovery. Applied Sciences 13(5): 3078. https://doi.org/10.3390/app13053078.
28. Zhang, R. H., Zhang, X. H., Camberato, J. J., and Xue, J. Q. 2015. Photosynthetic performance of maize hybrids to drought stress. Russian Journal of Plant Physiology 62: 788–796. https://doi.org/10.1134/S1021443715060187.
29. Meng, L.‑L., Song, J.‑F., Wen, J., Zhang, J., and Wei, J.‑H. 2016. Effects of drought stress on fluorescence characteristics of photosystem II in leaves of Plectranthus scutellarioides. Photosynthetica 54(3): 414–421. https://doi.org/10.1007/s11099‑016‑0191‑0.
30. Zhou, R., Kan, X., Chen, J., Hua, H., Li, Y., Ren, J., Feng, K., et al. 2019. Drought‑induced changes in photosynthetic electron transport in maize probed by prompt fluorescence, delayed fluorescence, P700 and cyclic electron flow signals. Environmental and Experimental Botany 158: 51–62. https://doi.org/10.1016/j.envexpbot.2018.11.005.
31. Liu, J., Li, H. J., Guo, Y. Y., Wang, G. X., Zhang, H. J., Zhang, R. H., and Xu, W. H. 2018. Effects of drought stress on the photosynthesis in maize. Russian Journal of Plant Physiology 65: 849.
32. Zhang, K., Chen, B‑h., Hao, Y., Yang, R., and Wang, Y‑a. 2018. Effects of short‑term heat stress on PSII and subsequent recovery for senescent leaves of Vitis vinifera L. cv. Red Globe. Journal of Integrative Agriculture 17(12): 2683–2693. https://doi.org/10.1016/S2095‑3119(18)62143‑4.
33. Zhang, L., Hu, T., Amombo, E., Wang, G., Xie, Y., and Fu, J. 2017. The alleviation of heat damage to photosystem II and enzymatic antioxidants by exogenous spermidine in tall fescue. Frontiers in Plant Science 8: 1747. https://doi.org/10.3389/fpls.2017.01747.
34. Fan, Q., and Jespersen, D. 2023. Assessing heat tolerance in creeping bentgrass lines based on physiological responses. Plants 12(1): 41. https://doi.org/10.3390/plants12010041.
35. Tan, W., Meng, Q‑W., Brestič, M., Olsovská, K., and Yang, X. 2011. Photosynthesis is improved by exogenous calcium in heat‑stressed tobacco plants. Journal of Plant Physiology 168: 2063–2071. https://doi.org/10.1016/j.jplph.2011.06.009.
36. Arslan, Ö. 2023. The role of heat acclimation in thermotolerance of chickpea cultivars: Changes in photochemical and biochemical responses. Life 13(1): 233. https://doi.org/10.3390/life13010233.
37. Mihaljević, I., Viljevac Vuletić, M., Tomaš, V., Vuković, D., and Zdunić, Z. 2025. Characterization of heat tolerance in two apple rootstocks using chlorophyll fluorescence as a screening method. Agronomy 15(6): 1442. https://doi.org/10.3390/agronomy15061442.
38. Kalaji, H. M., Carpentier, R., Allakhverdiev, S. I., and Bosa, K. 2012. Fluorescence parameters as early indicators of light stress in barley. Journal of Photochemistry and Photobiology B: Biology 112: 1–6. https://doi.org/10.1016/j.jphotobiol.2012.03.009.
39. Lee, J. H., Cabahug, R. A. M., You, N. H., and Nam, S. Y. 2021. Chlorophyll fluorescence and growth evaluation of ornamental foliage plants in response to light intensity levels under continuous lighting conditions. Flower Research Journal 29(4): 320–322. https://doi.org/10.11623/frj.2021.29.3.05.
40. Bayat, L., Arab, M., Aliniaeifard, S., Seif, M., Lastochkina, O., and Li, T. 2018. Effects of growth under different light spectra on the subsequent high light tolerance in rose plants. AoB Plants 10(5): ply052. https://doi.org/10.1093/aobpla/ply052.
41. Ceusters, N., Valcke, R., Frans, M., Claes, J. E., Van den Ende, W., and Ceusters, J. 2019. Performance Index and PSII connectivity under drought and contrasting light regimes in the CAM orchid Phalaenopsis. Frontiers in Plant Science 10: 1012. https://doi.org/10.3389/fpls.2019.01012.
42. Faseela, P., and Puthur, J. T. 2016. Chlorophyll a fluorescence changes in response to short‑ and long‑term high‑light stress in rice seedlings. Photosynthetica 54(3): 549–558.
43. Faseela, P., and Puthur, J. T. 2018. The imprints of the high light and UV‑B stresses in Oryza sativa L. ‘Kanchana’ seedlings are differentially modulated. Journal of Photochemistry and Photobiology B: Biology 178: 551–559. https://doi.org/10.1016/j.jphotobiol.2017.12.009.
44. Mlinarić, S., Antunović Dunić, J., Skendrović Babojelić, M., Cesar, V., and Lepeduš, H. 2017. Differential accumulation of photosynthetic proteins regulates diurnal photochemical adjustments of PSII in common fig (Ficus carica L.) leaves. Journal of Plant Physiology 209: 1–10. https://doi.org/10.1016/j.jplph.2016.12.002.
45. Lu, T., Yu, H., Li, Q., Chai, L., and Jiang, W. 2019. Improving plant growth and alleviating photosynthetic inhibition and oxidative stress from low‑light stress with exogenous GR24 in tomato (Solanum lycopersicum L.) seedlings. Frontiers in Plant Science 10: 490. https://doi.org/10.3389/fpls.2019.00490.
46. Li, L., Li, X.‑Y., Xu, X.‑W., Lin, L.‑S., Zeng, F.‑J., and Chen, F.‑L. 2013.
Assimilative branches and leaves of the desert plant Alhagi sparsifolia Shap. possess a different adaptation mechanism to shade.
Plant Physiology and Biochemistry 74: 239–245. https://doi.org/10.1016/j.plaphy.2013.11.009.
47. Liang, Y., Chen, H., Tang, M., Yang, P., and Shen, S. 2007. Responses of Jatropha curcas seedlings to cold stress: photosynthesis‑related proteins and chlorophyll fluorescence characteristics. Physiologia Plantarum 131(3): 508–517. https://doi.org/10.1111/j.1399‑3054.2007.00974.x.
48. Huang, X., Shi, H., Hu, Z, Liu, A., Amombo, E., Chen, L., and Fu, J. 2017. ABA is involved in regulation of cold stress response in Bermudagrass. Frontiers in Plant Science 8: 1613. https://doi.org/10.3389/fpls.2017.01613.
49. Mazur, M., Matoša Kočar, M., Jambrović, A., Sudarić, A., Volenik, M., Duvnjak, T., and Zdunić, Z. 2024. Crop‑specific responses to cold stress and priming: Insights from chlorophyll fluorescence and spectral reflectance analysis in maize and soybean. Plants 13(9): 1204. https://doi.org/10.3390/plants13091204.
50. Fan, J., Hu, Z., Xie, Y., Chan, Z., Chen, K., Amombo, E., Chen, L., and Fu, J. 2015. Alleviation of cold damage to photosystem II and metabolisms by melatonin in Bermudagrass. Frontiers in Plant Science 6: 925. https://doi.org/10.3389/fpls.2015.00925.
51. Li, J., Muneer, M. A., Sun, A., Guo, Q., Wang, Y., Huang, Z., Li, W., and Zheng, C. 2023. Magnesium application improves the morphology, nutrient uptake, photosynthetic traits, and quality of tobacco (Nicotiana tabacum L.) under cold stress. Frontiers in Plant Science 14: 1078128. https://doi.org/10.3389/fpls.2023.1078128.
52. Sun, L., Li, X., Wang, Z., Sun, Z., Zhu, X., Liu, S., Song, F., Liu, F., and Wang, Y. 2018. Cold priming induced tolerance to subsequent low temperature stress is enhanced by melatonin application during recovery in wheat. Molecules 23(5): 1091. https://doi.org/10.3390/molecules23051091.
53. Tsimilli-Michael, M. (2020). Revisiting JIP-test: An educative review on concepts, assumptions, approximations, definitions and terminology. PHotosynthetica 58: 275-292.

